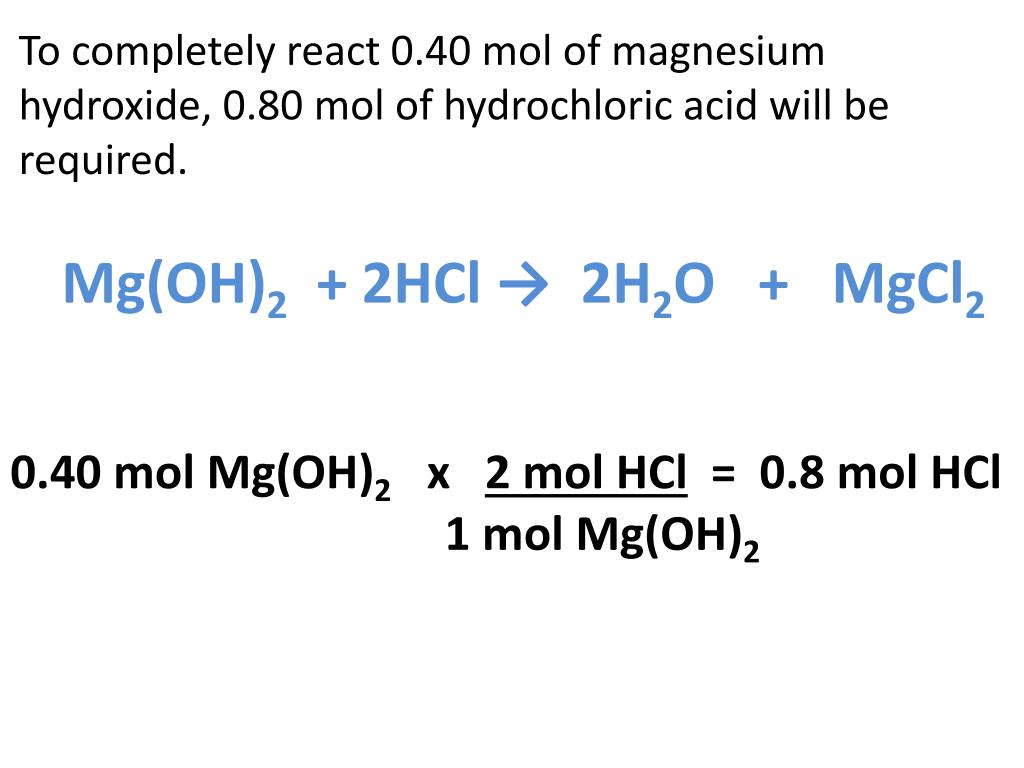
Mass of acetic acid x 100 = w/w% acetic acid Mass of vinegar 9. Use the masses from #5 and #7 to calculate the percent by weight (w/w%) of acetic acid in commercial vinegar. Assume commercial vinegar has a density of 1,00 g ml, use the volume of commercial visar from to calculate the mass of commercial vinstrated in sich samele MacBook Air Dll 다. Sample I moles acetic acid-M commercial vinegar sample 0.002500 L commercial vinegar 7. Remember 25.00 mL -0.02500 L of acetic acid were used and 1/10 of commercial vinegar is in each sample (0.002500L). Calculate the molarity of commercial vinegar each sample using the moles calculated for cach sample in #3. Calculate the mass of acetic acid for each sample using moles of acetic acid for each sample in #3 Moles of acetic acid x molar mass acetic acid - g of acetic acid 6. Calculate moles of acetic acid using moles of NaOH for each sample from #2 Remember 1:1 ratio of acetic acid to NaOH Sample I moles NaOH x 1 mols acetic acid - moles of acetic acid 1 mole NaOH 4. 
Calculate moles of NaOH for each sample moles NaOH sample 1 - Volume NaOH in L sample 1 * Molarity NaOH 3. Record the volume of NaOH used in each sample of vinegar (data table B), make sure to convert me to L. Pages > of 7 Mass of KHP Moles of KHP Moles of NaOH Volume NaOH used in liters Calculated NaOH molarity Part B: Titration of Vinegar: (Record all amounts in table B) 1. Calculate Average Molarity of NaOH (add up M of NaOH of all 3 samples and divide by 3): Table Part A: Sample 1 Sample 2 Sample 3 MacBook Air Calculate molarity of NaOH for each sample: M NaOH sample 1 - moles NaOH sample Volume NaOH in L sample 1 6. Record the volume of NaOH used in cach sample of KHP (data table A), make sure to convert mL to L. Remember 1:1 ratio of NaOH to KHP Sample 1 moles KHP x 1 mole NaOH 1 mole KHP 4. Calculate moles of NaOH using moles of KHP for each sample from #2. 
Calculate moles of each sample of KHP: Sample I mass KHP 1 mol KHP mol KHP Molar mass KHP 3. Standardization of Sodium Hydroxide Solution: (Record all amounts in table A) 1.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
